Planning a first launch requires unique timing considerations to ensure readiness and a successful launch.
Use this quick guide to learn about:
- Market pressures making this more challenging now
- How peers are planning their launch
- Common sequencing missteps and how to avoid them
- An example launch readiness timeline
- Tips for a successful product and company launch
MARKET PRESSURES
First-time launchers face tighter timelines and greater complexity
What’s making it harder now:
- Teams face capital constraints and pressure to do more with less
- Timelines are faster and more uncertain, with assumptions shifting mid-stream
- The market is changing at a rapid pace, resulting in reactionary responses
- Data and vendor proliferation increase choice and execution complexity
WHAT PEERS ARE DOING
More to build within the same planning horizon
Based on our Commercialization That Works research, the responses below reflect how industry leaders are currently approaching their launch timing and what drives confidence for emerging biopharma companies in their planning.
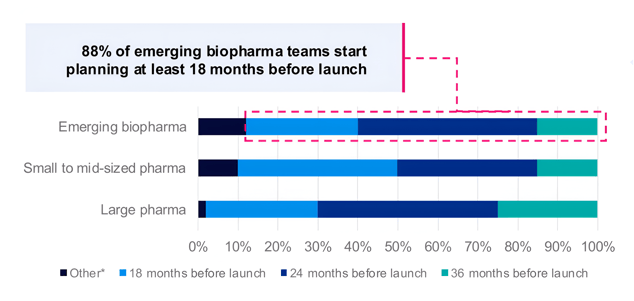
Source: At what timeframe relative to expected launch do you typically start planning for launch? n=120 respondents (n=40 each company size) *12 or 6 months before launch or it's a continuous process from launch to launch.
COMMON MISSTEPS
5 common first-launch sequencing pitfalls
| Common misstep | What breaks as a result | Recommended actions to avoid this |
| Planning how get to the finish line instead of "winning" | Teams default to activity over impact, checking tasks off the list without prioritizing what will drive launch success. | - Define strategic intent and a clear launch hypothesis early - Test assumptions with targeted market research and data - Align cross-functional activities to shared launch objectives |
| Relying on a single “launch date” |
Waiting for one all-encompassing go-live creates bottlenecks and last-minute scrambling. | - Define interim launch milestones across functions - Include a medical affairs data dissemination launch milestone - Tie data, systems, and governance to these checkpoints |
| Not using medical affairs as a bridge between clinical and commercial | Engagement strategy, insight capture, and evidence planning fall behind, and it's difficult to recover during launch year. | - Define medical launch timing alongside commercial - Build insight capture processes early - Align medical data flows before launch execution |
| Implementing core infrastructure during launch year due to cost concerns | Waiting until launch year to implement ERP, G&A systems, or core operational platforms complicates integrations and uses launch bandwidth. | - Implement and stabilize major systems 12-18 months before launch - Validate distribution/hub/claims feeds and reporting logic before approval - Protect launch bandwidth by stabilizing core operations early |
| Focusing on perfecting systems rather than on what’s needed for launch | Teams become focused on the implementation rather than the launch, and the company build becomes a distraction to the launch. | - Sequence foundational capabilities before optimization capabilities - Set expectations early about what will and won’t be ready at launch - Prioritize investments that directly protect launch visibility |
SEQUENCING IN ACTION
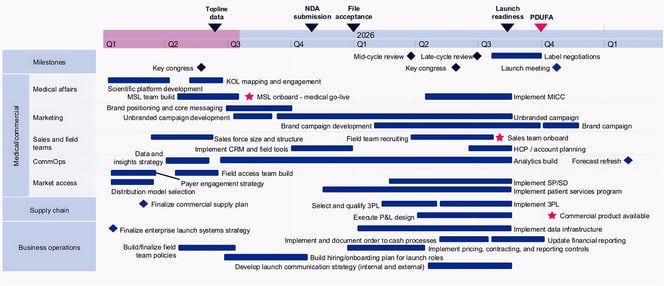
Example launch readiness timeline
This sample timeline illustrates how first-time launch activities could be staged across functions to protect launch readiness while building foundational capabilities in parallel.
SUCCESSFUL LAUNCH
Establish priorities and phase the rest
Launching a product and building a company at the same time requires more than early planning. It relies on clear expectations and disciplined staging to ensure launch readiness drives real performance and the greatest chance of "winning."
-
Prioritize the right activities to maximize launch impact.
Align teams around the specific objectives you intend to achieve and concentrate resources on the priorities that will move the needle toward success.
-
Position medical affairs as an early medical and scientific community builder.
Coordinate activity around publications, conference strategy, supporting materials, and your CRM for informed scientific engagement with HCPs and KOLs pre-launch.
-
Create interim milestones.
Tie systems, hiring, governance, and cross-functional work to phased checkpoints to prevent launch-year compression.
-
Focus on launch readiness rather than system completeness.
Set expectations early on what will be ready at launch, what will phase post-launch, and what the IT team can realistically build and sustain.
-
Set clear criteria before engaging with vendors.
Take the time to document requirements and selection criteria first to avoid committing to more than you need and can operationalize.
For more data vendor-related insights in life sciences, read our article: Building a vendor strategy: Protecting commercial plans from supplier-driven data risk